|
Logically, this should be the person making the product available on the EU single market.Ĭontents of the Declaration of Conformity However, the CE directives don’t necessarily specify who has an obligation to translate it. The DoC must be drawn up in one of the official languages of the European Union. The Declaration of Conformity must be kept for ten yearsįrom the date of placing the last product on the market unless the relevant CEĭirectives provide for any other duration. If the Declaration isn’t available or is invalid, the market authorities will recall the product, issue a monetary fine and the person who placed the product on the market could face imprisonment. In most cases, when a product is covered by several product safety directives, only a single DoC needs to be drawn up.Ī copy of the Declaration of Conformity should be added to the technical file and made available upon request of the national market surveillance authorities. 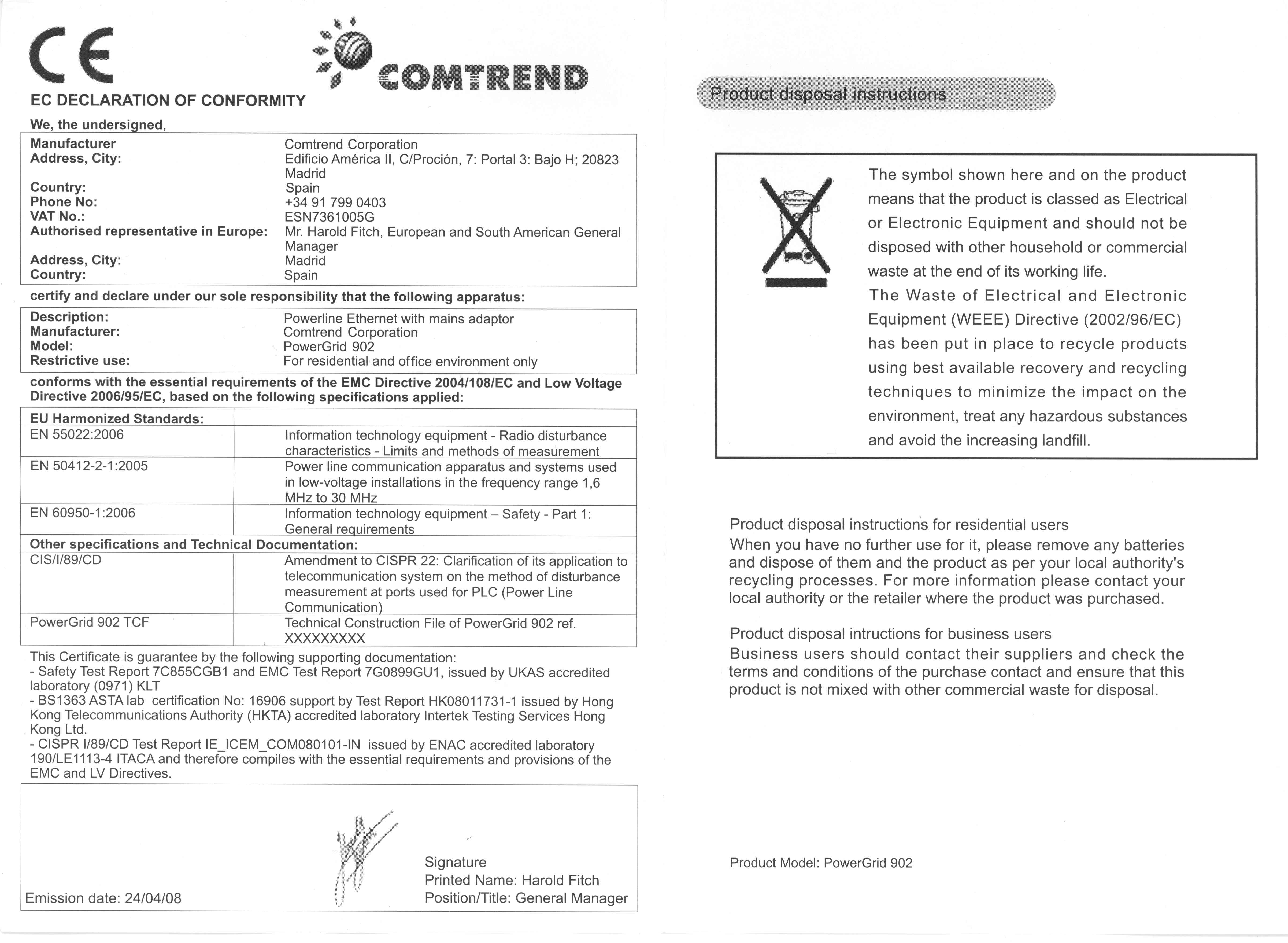
The Declaration of Conformity is mandatory for any product that falls within the scope of the CE marking legislation. The DoC is drawn up only after the technical documentation (aka technical file) The manufacturer assumes full responsibility for the product’s CE compliance. By drawing up and then signing the EU Declaration of Conformity,  
Wherein a manufacturer formally declares the compliance of a product with theĮssential health, safety and environmental requirements of the relevantĭirectives. The EU Declaration of Conformity (DoC) is a legal document,
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
